Question #13: Two applications of transgenic animals and knock-out mice
Here's a quick rundown of transgenic animals: By deleting a particular gene or a particular promoter or some aspect of the genetic code, researchers can determine the role of that particular gene. Transgenic knock-outs can be for one or both of the gene alleles at a given locus.
These days the name of the game is time- or tissue-specific gene knock-outs. This allows for the deletion of your favorite gene only when you the experimenter manipulate it. In this manner, you can logically determine the function of genes whose universal knock-out might prove lethal or in some other way mess with normal development. The two most publicized uses of conditional knockouts are in optogenetics and tissue-specific imaging, and I'll give recent examples of each:
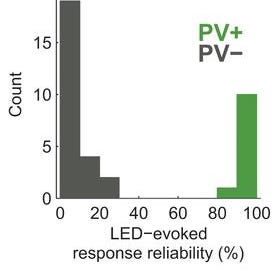
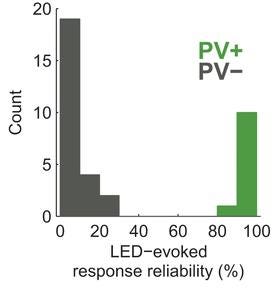
1) Identifying neuron types in vivo: One way of using channelrhodopsin-2 is to express it only in certain neurons so that neuron type can be identified via extracellular recording when blue light is flashed, based on the presence or absence of a short latency action potential. Lima et al used this method with transgenic mice that expressed the gene Cre recombinase driven by the parvalbumin promoter, which is expressed in many interneurons.
These researchers then inserted a viral vector (AAV) containing a transcriptional insulator flanked by two loxP sites (loxP-STOP-loxP), downstream of the cytomegalovirus promoter into their mice and upstream of the genes for channelrhodopsin-2 (ChR2) / yellow fluorescent protein. In cells expressing Cre recombinase (i.e., the same cells that express parvalbumin) and in which the virus successfully enters, Cre excises the STOP sequence and allows for the expression of ChR2.
The system allows the determination of whether or not a given cell is of a type (i.e., a parvalbumin-expressing interneuron) via just extracellular recording, because light will cause a ChR2-dependent action potential in those cells. The reliability of cells to respond to light activation (LED) with short AP's as a result of optogenetics never ceases to amaze me:
2) Color timer mice: Livet et al's brainbow study (here) has been cited 100+ times in the 2.5 years since it's been published, so you've all heard of that form of tissue-specific imaging and now it's boring. Building on that technique is Kanki et al's strategy to image the differentiation of neural stem cells into adult neurons.
First, they inserted the gene for an orange fluorescent protein after the promoter for the intermediate filament protein Nestin, which is expressed specifically in neural stem cells. The gene will be present in all cells of the mice but will only be expressed in neural stem cells, and since the protein it makes is fluorescent the researchers can tell whether the cell is a neural stem cell. To test the effectiveness of the genetic implant they measured the correlation between an antibody for Nestin (alexa488) and the orange fluorescent protein (KOr) in dissociated neural cells, and found it to be linear and positive:

They then developed transgenic mice with gene fluorescent protein driven by the promoter for doublecortin, a microtubule-associated protein expressed in immature neurons. Next, they crossed the two type of transgenic mice to get double tissue-specific transgenic mice. This allowed them to visualize the transition from neural stem cell to neurons between the subgranular zone and olfactory bulb (the rostral migratory stream) of adult mice:

As you can see, orange fluorescent protein (KOr) becomes less distinct along the path as green flourescent protein (EGFP) becomes more distinct, as expected during neuronal differentiation.
The disadvantage of this technique is that either you have to breed the animals to have the transgene or you have to insert it in vivo, which is problematic both in targeting the right cells, getting the DNA vectors inside the cells, and getting the genes to incorporate into the genome properly. And if you know how to do the latter effectively, don't bother with this stuff... go collect your Nobel already.
Inspired by CalTech’s Question #13 for cognitive scientists: “What is a transgenic animal? A transgenic knockout mouse? What are the advantages and disadvantages of such animals for neuroscientific studies? Please give one or two specific examples.”
References
Kanki H, et al. 2010 "Color Timer" mice: visualization of neuronal differentiation with fluorescent proteins. doi: 10.1186/1756-6606-3-5.
Lima SQ, Hromádka T, Znamenskiy P, Zador AM (2009) PINP: A New Method of Tagging Neuronal Populations for Identification during In Vivo Electrophysiological Recording. PLoS ONE 4(7): e6099. doi:10.1371/journal.pone.0006099